One-day cervical cancer test could soon be available in South Africa

Cervical cancer kills more South African women than any other type of cancer and one in every 39 women in the country will be diagnosed with it in her lifetime.

Cervical cancer or its precursor, abnormal cervical cells, are generally detected using a pap smear test – where cells are lifted from the cervix using a swab and sent to a laboratory for testing. If abnormal cells are detected, a biopsy has to be done in which a small tissue sample is cut from the cervix and sent back to the pathologist for further testing.
Depending on these results, the woman will receive treatment or not. This process can take up to several weeks and requires a woman to make three trips to her health care provider.
“You can lose a lot of patients through follow-up, and the best method is to do everything in one visit,” said Kathleen Schmeler, an associated professor at the MD Anderson Cancer Centre at the US University of Texas.
She added that in many African countries, a lack of laboratories and pathologists has also limited women’s access to pap smears.
Schmeler presented a new devise, called the high-resolution microendoscope (HRME), which could provide women with same-day pap smear results without the need for laboratories at the AORTIC International Cancer Conference in Durban last week.
“With the HRME everything can be done in one day, and you no longer need a specialist and a lab and a pathologist,” said Schmeler, who was part of the team of researchers at MD Anderson’s Department of Gynaecologic Oncology and Rice University’s Department of Bioengineering that developed the HRME.
The device consists of a digital florescent microscope, a fibre optic probe, a cell phone and battery pack. The probe is placed on the cervix and transmits images of cells to the cell phone screen. Normally, pathologists only see these types of images beneath a microscope.
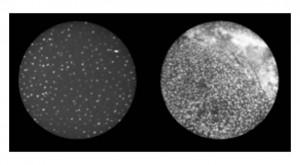
“You can easily identify irregular (cells) by their shape and how close they are together,” Schmeler told Health-e. “To remove the element of human error, (the HRME) also comes with software that does the calculations and tells you if (cells are) normal or not.”
Costing about R5 000, the HRME could potentially bring relief to overburdened pathology services in South Africa and other African countries. The United States has one pathologist for every 20 000 patients, but in South Africa there are only one for up to every 500 000 patients. In other African countries, there may be just one pathologist serving several million people.
The HRME has shown promising results in several small and medium pilot studies, and large studies are planned for South Africa soon.
“We are very optimistic about this new technology,” said African Cancer Institute Director Vikash Sewram. “This is exactly what we need at primary health care facilities—a point of care test.”
Sewram said he believes the devise may not only be useful in screening for cervical cancer, but also oesophageal cancer, for which there is currently no form of screening.
“There is a lot of benefit to this technique, we just need to validate it to ensure that in our population it can work just as well as the current gold standard, the pathological diagnosis,” he added. “I see no reason that if it does stand up to the test that it will not be rolled out in the country.” – Health-e News Service.
Author
Republish this article
This work is licensed under a Creative Commons Attribution-NoDerivatives 4.0 International License.
Unless otherwise noted, you can republish our articles for free under a Creative Commons license. Here’s what you need to know:
You have to credit Health-e News. In the byline, we prefer “Author Name, Publication.” At the top of the text of your story, include a line that reads: “This story was originally published by Health-e News.” You must link the word “Health-e News” to the original URL of the story.
You must include all of the links from our story, including our newsletter sign up link.
If you use canonical metadata, please use the Health-e News URL. For more information about canonical metadata, click here.
You can’t edit our material, except to reflect relative changes in time, location and editorial style. (For example, “yesterday” can be changed to “last week”)
You have no rights to sell, license, syndicate, or otherwise represent yourself as the authorized owner of our material to any third parties. This means that you cannot actively publish or submit our work for syndication to third party platforms or apps like Apple News or Google News. Health-e News understands that publishers cannot fully control when certain third parties automatically summarise or crawl content from publishers’ own sites.
You can’t republish our material wholesale, or automatically; you need to select stories to be republished individually.
If you share republished stories on social media, we’d appreciate being tagged in your posts. You can find us on Twitter @HealthENews, Instagram @healthenews, and Facebook Health-e News Service.
You can grab HTML code for our stories easily. Click on the Creative Commons logo on our stories. You’ll find it with the other share buttons.
If you have any other questions, contact info@health-e.org.za.
One-day cervical cancer test could soon be available in South Africa
by wilmastassen, Health-e News
November 28, 2013
MOST READ
FS woman waits 4 years for medical negligence pay out after ‘out of court settlement’
Prolonged power outage leaves hospitals in the dark for two days
There’s more to self-care than scented candles or massages, it’s a key public health tool
Access to clean water and stable electricity could go a long way to addressing rising food poisoning in SA
EDITOR'S PICKS
Related


Patents could block access to breast cancer treatment for decades

